Inside this newsletter:
1. Food Science Factoids: a bit about Freeze Drying!
2. When to see me?
3. Deals!
(That title is a chemistry-nerdy-witty-clever pun, just FYI. Keep reading if you want to know why! And keep reading even if you know why!)
I love this newsletter because it challenges me to explain concepts that I think I fully understand! I always learn something new! This particular newsletter is no exception!
Folks often ask me about my products, “so this is freeze dried?”
No… my K9 Bros and dried fruit are dried using a "dehydrator", which technically should be called an "air convection dryer" or a "Cabinet" or "Tray Dryer" (but everyone calls my machine-type a "dehydrator.") The other thing I hear is “I have a food dehydrator, too.” And then in my ego-manliness (insert accurate name of human psychological pathology here) I show a picture of my massive dehydrator. I’d say mine is bigger than yours (bordering on toxic masculinity, but I’d simply say this is a few degrees before: simple-ignorant manliness.)
What's intriguing about "freeze drying" is the name itself is an oxy-moron. Freeze drying is also about as food-sciency as it gets, so I thought I’d do a quick explanation of what is freeze drying.
So first, my title is so awesome “Freeze Drying is Quite Sublime” since "sublime" besides being defined as “characterized by nobility; majestic, of high spiritual, moral, or intellectual worth”, to sublime (or “sublimation”) is the process of changing from solid directly to vapor. (alternative to evaporation from liquid to gas)
A great common example of sublimation is dry ice, which is solid-frozen carbon dioxide. CO2 has been frozen below -109 F Or -78.5 C, which is really cold! Dry-ice is also wonderful as a lightweight coolant in place of frozen water ice, which gets sloshy/is a mess, is heavy, and can contaminate the food you’re trying to keep frozen in a portable cooler.
So freeze drying takes advantage of the phenomenon of sublimation (the change of state from solid ice to gas-water-vapor.)
So why is freeze-drying so great of a process? Well, it’s arguable the best food preservation method for maintaining raw nutritional properties and a lightweight, porous texture that rehydrates rapidly.
If you want to sterilize the food and kill pathogens, or cook the food slightly, then dehydration with heat in the range of 115F to 150F is better than freeze-drying. (dehydration with heat: the temperature selection depends on the food, be it a fruit, vegetable, or meat. But that's a different topic.)
A great example of a food candidate for freeze drying is a strawberry: you've seen freeze dried strawberries in a box of cereal. A dehydrated strawberry (by using heat) might become very hard / shrunken, and you could hurt your teeth chewing on it, whereas a freeze dried strawberry can be munched down quickly, without even time needed for your milk to rehydrate the strawberry (too much time and your cereal flakes might become soggy.)
So how does water sublime? Well, here’s my cheat sheet. (phase diagram of water with notations)
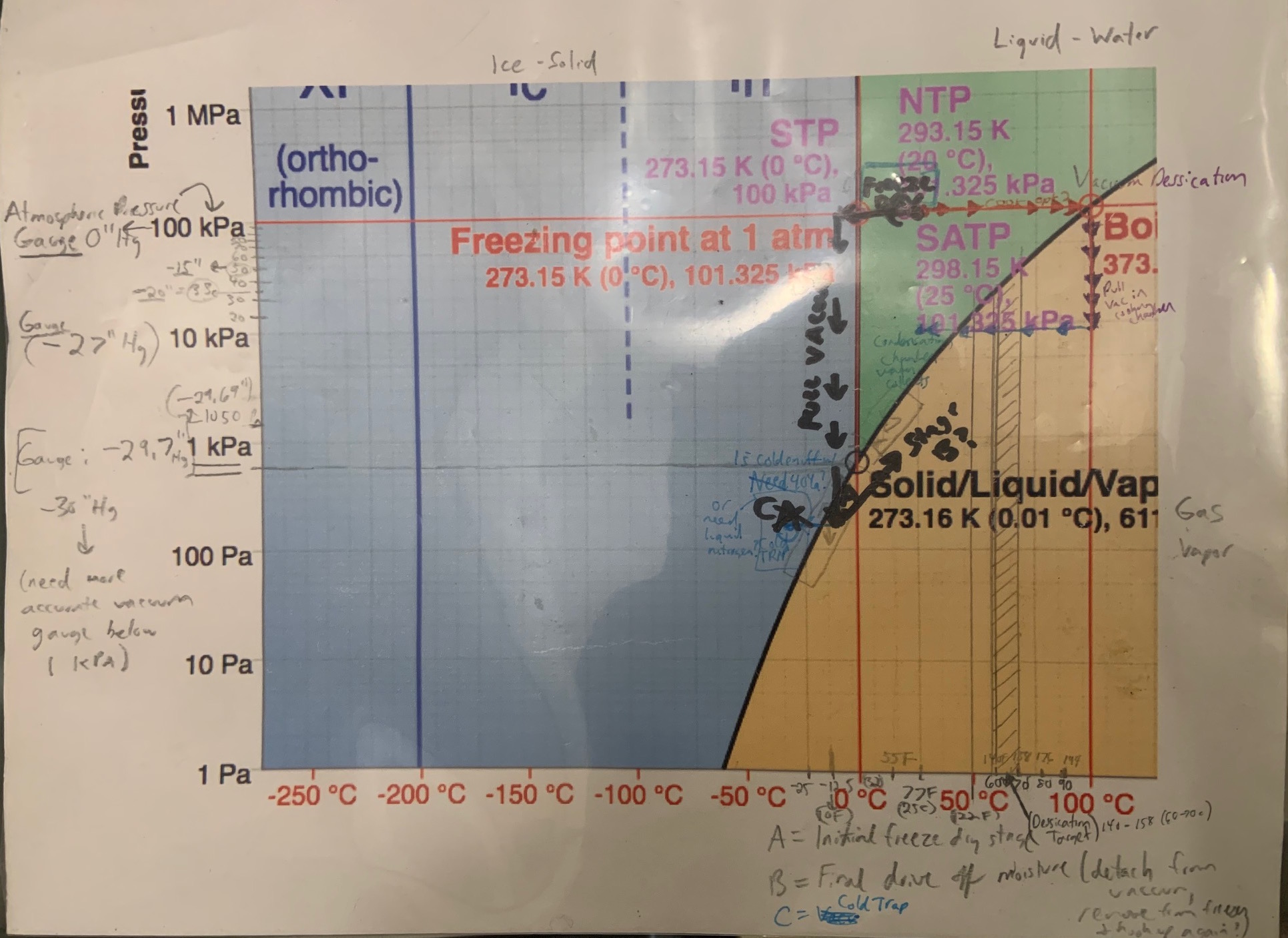 Water sublimes when the pressure drops to approximately 0.62% of atmospheric pressure!!! at 0 Celsius (32F.) The scales for pressure, atmospheric pressure: 1 “atm” = 14.7 psi = 760 mm Hg (Mercury pressure gauge.) The Mercury scale must read below 4.7mm Hg!
Water sublimes when the pressure drops to approximately 0.62% of atmospheric pressure!!! at 0 Celsius (32F.) The scales for pressure, atmospheric pressure: 1 “atm” = 14.7 psi = 760 mm Hg (Mercury pressure gauge.) The Mercury scale must read below 4.7mm Hg!
Most machines when running have a vacuum gauge, typically measured in mm Hg and is a negative value. Close to -30mm Hg is a “perfect vacuum” although at extremely high vacuums, the unit of measurement used are Pascals.
The two new things I learned: previously, I had read that during freeze drying, the food is heated slightly (despite-still maintaining the frozen-ice within the food, which is hard to fathom.) ***The sublimation of solid ice to vapor keeps the food frozen despite heat being applied! Let me elaborate:
First, the food is sandwiched in between a mesh which allows for vapor to escape. Outside the mesh is a heated plate which heats the dried portion of the food. The dry food is porous, allowing the inner portion of the food, still containing ice, to sublime, from the surface of the ice (the "ice front"), outwards. The moisture can sublime faster than the ice can melt. (rate of heat mass transfer vs rate of heat transfer)
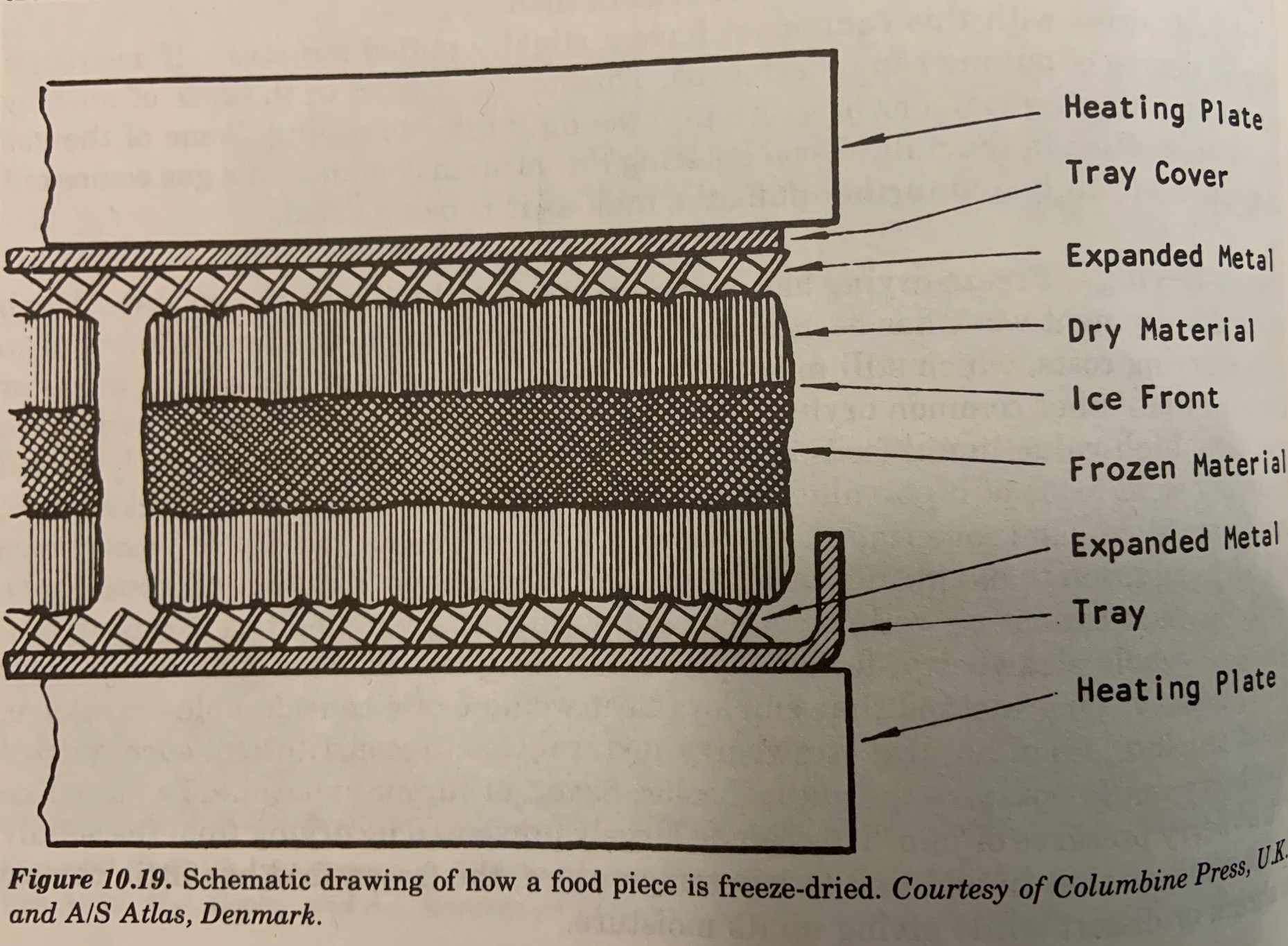
Secondly, as the water sublimes, the water molecules of gas absorb energy (known as the “latent heat of sublimation", similar to how you get very cold when you sweat, the water on your body is evaporating to a gas, known as the “latent heat of vaporization.”
I thought that was quite “cool.” (another great pun!)
Ok, I’m going to quit while I’m ahead. There are other complexities to freeze drying (eg: the vacuum pressure) which requires a part 2!
ii. New batch of BLACK GARLIC!
(amazingly delicious AND nutritious!)
Farmers Market special: 2nd bulb, $1 off!
2. When to see moi next?
Answer: This weekend! Here's the full schedule:
|
Saturdays |
Sundays |
|
| This weekend! |
Glen Cove Nov 12th 10am - 2pm (1 hour later start) |
Huntington Nov 13th 7:30am - noon |
More info & directions:
Glen Cove-Deep Roots Farmers Market

3. Deals!
Subscription direct to your door!
If you can’t come to my products, make my products come to you!
Subscribe and Save! 5% off and free shipping on qualifying orders!
Shameless plugs
Fresh dried fruit is a weight loss cure!
(this statement that has not been substantiated by any scientific study... but no sugar added dried apples and pears can't be bad!)
No sugar added! USA grown! New York State Red Apples! Slow and low dehydrated to preserve vitamins, antioxidants, and flavors!
(compare to other brands apple chips which are high-temperature baked)
1) Red Apples!
2) Green Apples!
3) Pears!
4) Nectarines!
5) Hmmm... what's next?

Order more wands or get a refill! Get your fill of bubbles this summer!
Conclusion:
Hypothesis: I will see you this weekend! 😃
Garvies Point tomorrow/Saturday: 1 hour later start: 10am start until 2pm
Huntington Sunday: 7:30am to noon!
Null hypothesis: I will not see you this weekend 😭
Conclusion: Not sure! Let's see!
![]()
★ k9bros.com ★
☆ gourmet-magic ☆
★ bubble-science ★










.png)